Liquid Crystalline Polymers
Photorefractivity of Isotropic Phase of LC polymers
Figure 1
The photorefractive effect of LC polymers was reported in 1999. The photorefractivity
of a LC polymer NBA6 (Figure 1) doped with 30 wt% DEH and 1 wt% TNF was
investigated. The photoconductive dopant, DEH, is not a liquid crystal,
so that the addition of 30 wt% DEH disturbed the liquid crystal phase of
the host polymer. The sample film was isotropic and transparent. The sample
film was sandwiched between two ITO-glass plates and the film thickness
was adjusted to 100 micro-meter. A typical example of the asymmetric energy
exchange in a two-beam coupling experiment is shown in Figure 2. The transmitted
intensities of the laser beams through the sample film are indicated as
a function of time. An electric field of 30V/micro-meter was applied to
the sample. When an electric field was applied to the sample, the transmitted
intensity of one beam increased while that of the other beam decreased.
A diffraction efficiency of about 40% was observed in this case. When the
field was turned off, the intensities of the two beams gradually returned
to their original values because of the relaxation of D-pai -A chromophores.
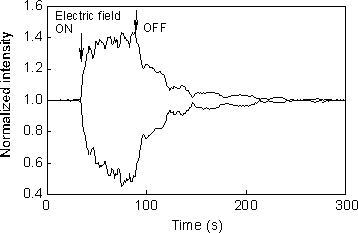
Figure 2
Effect of Liquid Crystallinity
The photorefractivities of liquid crystalline polymers were compared with
that of amorphous polymers. Ac-NBA6 did not exhibit a liquid crystal phase
even though the molecular structure is almost the same as that of NBA6.
The only difference is the structure of the main chain. NBA6 is the methacrylate
polymer and Ac-NBA6 is the acrylate polymer. The diffraction efficiencies
of Ac-NBA6, methacrylate NBA6 and NBA3 are plotted as a function of the
applied electric field in Figure 3. The two-beam coupling experiments were
conducted under the condition of a transparent isotropic phase of the polymers
at room temperature. The concentration of DEH was 30 wt% and TNF was 1
wt%. The diffraction efficiency of Ac-NBA6 was found to be much smaller
than that of NBA6, although the molecular weight was almost the same for
these polymers and the glass transition temperature of Ac-NBA6 was lower
than that of NBA6. The diffraction efficiency of amorphous polymer NBA3
was also much smaller than that of NBA6. Thus, the photorefractivity is
larger in the isotropic phase of the liquid crystalline polymer compared
to that of the amorphous polymers. The photorefractive properties of the
cyanobiphenyl PLCs were also investigated.30 CB6 and CB8 exhibit both nematic
and smectic phases. CB3 exhibit a nematic phase; however, the order parameter
was very small. CB3 is clearly inferior to CB6 and CB8 in terms of the
ability to form a liquid crystal phase. The diffraction efficiency of CB3
was several times smaller than those of CB6 and CB8, although the CB3 sample
was transparent and the glass transition temperature of CB3 was almost
the same as that of CB6. A 1:1 copolymer of methylmethacrylate and 4-methacryloyloxyhexyloxy-4'-
nitroazobenzene (NitroAzo6) exhibits a nematic liquid crystal phase, whereas
a 1:1 copolymer of methylmethacrylate and 4'-methacryloyloxyhexyloxy-2'5'-dimethyl-
4-nitroazobenzene (DMAzo6) does not exhibit a liquid crystal phase. The
diffraction efficiencies of polymers NitroAzo6 and DMAzo6 mixed with photoconductive
compounds (CDH and TNF) were investigated. The diffraction efficiency of
DMAzo6 was found to be much smaller than that of NitroAzo6, although their
molecular weights are almost the same and DMAzo6 exhibits a lower glass
transition temperature than that of NitroAzo6. It can be concluded that
the photorefractivity becomes large in the isotropic phase of the liquid
crystalline polymer in comparison with the polymers with low liquid crystallinity.
These results indicate that the isotropic phase of liquid crystalline polymers
is not the same as that of the amorphous polymers. As the molecular structure
of NBA6 is almost identical to that of Ac-NBA6, the enhancement of photorefractivity
may arise from the morphology of the isotropic phase. The isotropic phase
of a LC is believed that the orientations of mesogens are completely random.
However, in the case of the isotropic phase of NBA6/DEH mixture, the orientations
of mesogens are randomized not by rising temperature, but by lowering of
the phase transition temperature through the introduction of an impurity
(DEH) into the LC polymer. The formation of an ordered structure is thermodynamically
prevented by the presence of the impurity. However, the local concentration
of DEH is not unity throughout the highly viscous medium like polymer film.
Thus, some fractions of mesogens tend to form locally ordered structure
in the microscopic region. It is likely that the electro-optic effect was
enhanced by the presence of the microdomain structure leading to larger
photorefractivity.
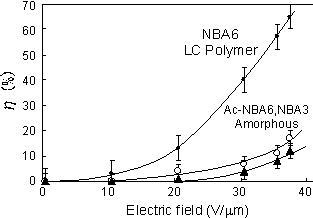
Figure 3
T. Sasaki, M. Goto, Y. Ishikawa and T. Yoshimi, J. Phys. Chem.
B, 103, 1925-1929 (1999).
T. Sasaki, R. Kai, A. Sato, Y. Ishikawa and T. Yoshimi, Mol. Cryat.
Liq. Cryst., 373, 53-70 (2002)
T. Sasaki, T. Shimada, K. Tachibana, Chem. Lett., 324-325
(2002)